Tracing the Scientific Breakthroughs Behind AKNETIDE®
AKNETIDE® R&D has been grounded over a decade of research from molecular discovery to clinical validation. Below is a snapshot of key scientific and regulatory milestones that demonstrate its journey from lab bench to real-world acne care efficacy.
2014
Inception of AMP
Inception of antimicrobial peptide discovery from nature and lab development
2019
Granted the world first ever AMP additive for animal nutrition and health
Granted the world first-ever antimicrobial peptide zootechnical additive for animal nutrition
2023
The cyclopeptide was discovered
AKNETIDE®, a novel antimicrobial cyclopeptide with potent inhibition against C. acne and S. aureus, was discovered
2024
First clinical study by SGS
The first-ever clinical study, with SGS Cosmetic Lab, was completed and demonstrated the significant efficacy of sensible acne level alleviation
2025
Multi-MOAs established and a strict hospital clinical study completed
Mechanism Elucidation: Multi-MOAs were established and proven by a leading cosmetic research institute
A double-blinded placebo-controlled Clinical Study in a world-famous Dermatology Hospital was conducted.
Scientifically proven and Dermatologist Endorsed evidences behind AKNETIDE®
Backed by 10+ years of rigorous consistent research of antimicrobial bioactive peptides, and recently validated by leading independent biochemical laboratories and top-tier dermatology hospital, AKNETIDE® harvested myriads of evidences, either of mechanism or of clinical studies, enables it deserving a worldwide breakthrough anti acne cosmetic ingredient designed for EFFICACY without bragging.
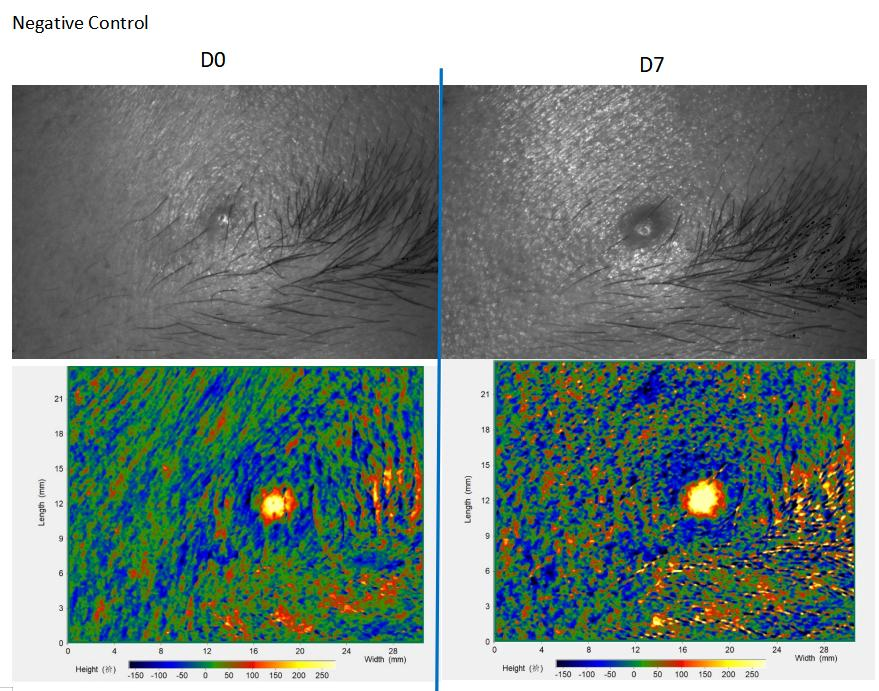
Double-Blind Hospital Study
A dermatology hospital clinical study. A 7-day double-blinded placebo-controlled clinical study demonstrated the prototype formula embedded ANKENTIDE® efficacious in alleviating acne and improving skin conditions of volunteers
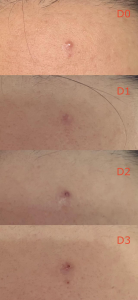
SGS Clinical Study
AKNETIDE® embedded prototype formula demonstrated potent efficacy in significantly alleviating volunteers’ acne conditions from black heads to papules in 7 days.
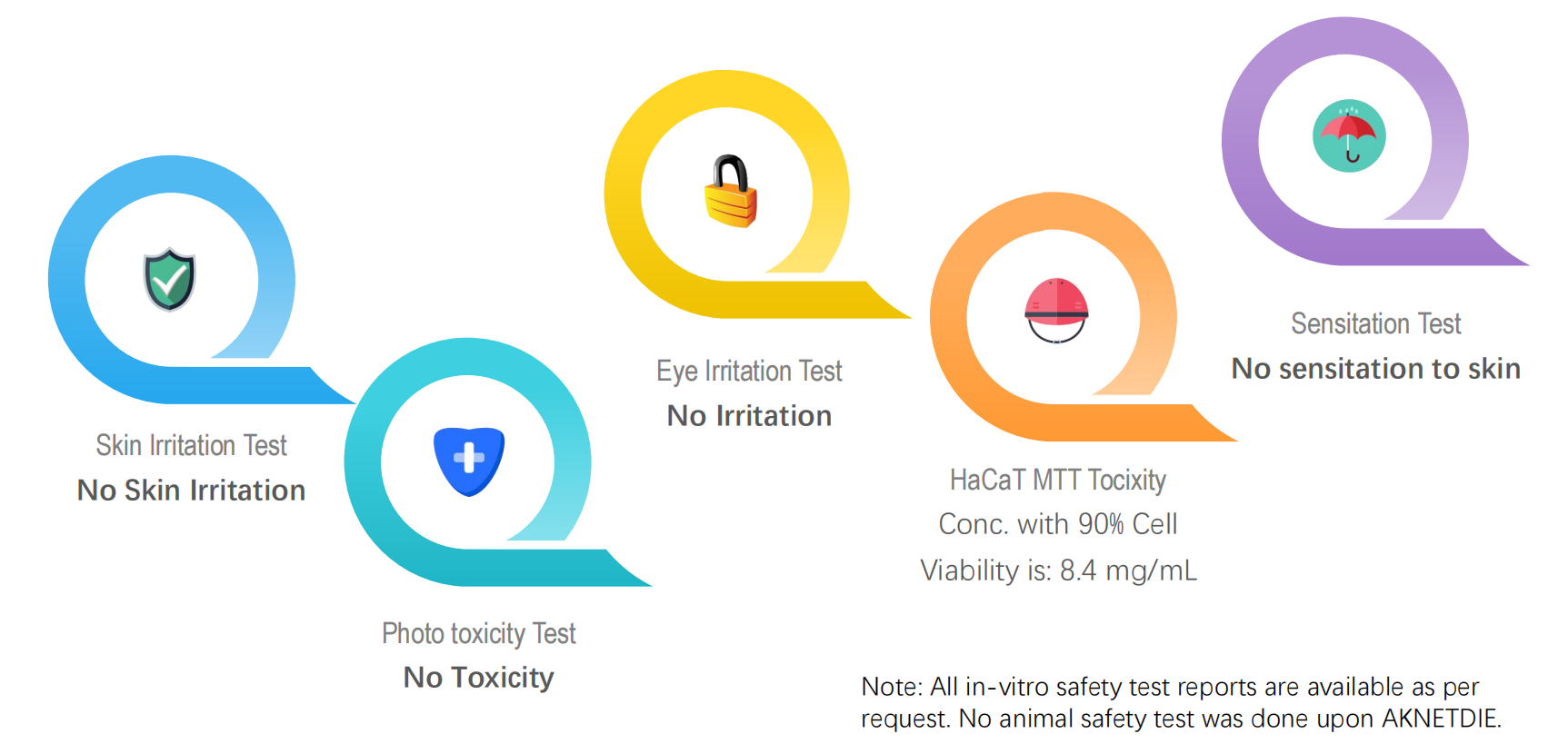
In-vitro Safety Studies
Skin irritation, phototoxicity, eye irritation, sensitization to skin were all tested NEGATIVE; HaCaT MTT Toxicity data was given.
Published Studies and Patents
Scientific publications documenting AKNETIDE®‘s clinical findings and mechanism of action are currently in preparation for peer-reviewed journal submission.
Pre-publication data is available to qualified cosmetic manufacturers and researchers under NDA.
Contact info@dermatide.net to request access.
Frequently Asked Questions
AKNETIDE® has been evaluated in a 7-day double-blind clinical study conducted through SGS, a globally recognised testing institution. Results showed an 88.7% reduction in blackheads, 51.0% reduction in papules, and 16.3% overall reduction in total acne lesions. A hospital-based dermatology trial was completed in 2025 to further validate multi-mechanism performance.
Yes. AKNETIDE® has undergone in-vitro safety assessments for irritation, phototoxicity, and sensitisation, all of which confirmed a favourable safety profile. It has received dermatologist-tested designation and is COSMOS-approved as a natural raw material — suitable for use in cosmetic formulations targeting sensitive and reactive skin types.
An antimicrobial cyclopeptide is a ring-structured peptide that mimics the body’s host defence mechanisms to selectively inhibit pathogenic bacteria. Unlike antibiotics or chemical actives, cyclopeptides work by disrupting bacterial membrane integrity without promoting antibiotic resistance. AKNETIDE® is derived from a natural cyclopeptide scaffold and is the first of its class developed for cosmetic acne applications.
Publication of AKNETIDE® ‘s clinical and mechanistic data in peer-reviewed journals is in progress. The foundational antimicrobial peptide research behind AKNETIDE® spans from 2014, with the first regulatory approval of a related AMP zootechnical additive in 2019. Scientific publications are expected to be available shortly — please contact info@dermatide.net for pre-publication data on file.